Publishing a paper as the sole author seems to be a rite of passage for any respectable scientist, and I’ve finally crossed the threshold!
Hannah, W. M., 2017: Entrainment vs. Dilution in Tropical Deep Convection. J. Atmos. Sci., 74, 3725–3747.
I started this work back in graduate school, but I had to put it on the back burner during my first postdoc. When I finally got back to working on this I realized that there were some big problems with my approach. I went through several rounds of review to work out all those kinks, but luckily the reviewers provided lots of good feedback. Part of this process involved rerunning the simulations and re-building the analysis code more times than I can remember. But I eventually ended up with a finished product I feel good about.
There were several things that got me thinking about this subject, but the main motivation came from reading papers about clouds and seeing the word “dilution” thrown around without any attempt to define or quantify it. We all have an intuitive sense of what “dilution” means, but can we measure it? Specifically, can we estimate the dilution rate of a cloud due to entrainment?
What is Entrainment?
verb – to draw in and transport by the organized current of a fluid
The word entrainment can also mean “to board a train”, which is a decent analogy for how it’s used in atmospheric science (the train in this case would be a cloudy updraft). Notice there is nothing in the definition of entrainment about dilution, but the words are often used interchangeably because the air that is being entrained is generally drier than the air inside the cloud, so we intuitively expect dilution. However, this is not always the case since the air being entrained can sometimes be relatively humid air that was previously detrained by a cloud.
What is Dilution?
noun – the action of making something weaker in force, content, or value.
In the most general sense for our context of fluid mechanics, dilution is the process of reducing the concentration of a substance. An undilute substance is in it’s purist state (i.e. highest concentration). Conversely, a completely dilute substance is mixed with other substances such that it is unrecognizable from the undilute state (i.e. low concentration).
There are a few things I could find online about calculating a dilution rate, like this Wikipedia article, but these are too simplified to apply to a cloud. The term is also used in finance, but that isn’t very helpful here.
A measure of dilution rate needs to produce a rate of zero if identical substances are mixed together, and should also allow for negative dilution (i.e. concentration) if a more concentrated version of the substance is added. So to describe dilution we need to know about the “dilutee” (substance being diluted) as well as the “diluter” (process doing the dilution). Additionally, a measure of dilution rate needs to account for the mass of each substance, since increasing the amount of the “diluter” should also increase the rate of dilution.
For now, consider a simpler problem that’s slightly more complicated than diluting an ambiguous chemical with some inert water. Imagine you want to describe the dilution of a cup of coffee as you add milk to it. First we need to know about the properties of the coffee.

- How is the concentration of coffee defined?
- by the concentration of caffeine?
- by the concentration of acids
- by the concentration of other chemicals that define the “flavor” of the coffee?
Each of these properties may be diluted at different rates, and they all might be important. The dilution rate of each property may also change if we change the properties of the milk being added. For instance, whole milk with a higher fat content will neutralize more acid than skim milk, but have the same effect on the level of caffeine. Also, the amount of coffee we start with and the rate at which we add a non-coffee substance will affect the dilution rate as well.
Dilution of a Cloud
So how do we apply all this to estimate the dilution of a cloud by entrainment?

First we need to decide on a cloud property Φ to use for measuring dilution. Then we need to write down a basic equation that describes the behavior of this variable and manipulate it to get an equation that describes the change in concentration over the volume of the cloud. I’ll skip the algebra, but essentially we end up with a ratio between a number describing the effect of entrainment and the anomalous value of Φ. Both of these quantities are summed over the volume of the cloud in order to consider the effects on the whole cloud and how that volume might be changing. The resulting dilution rate calculation looks something like this:
![]()
The units of the dilution rate are seconds-1, so the dilution rate is like an exponential decay rate or an e-folding timescale. If we invert the dilution rate we get the time it takes to reduce the anomalous value of Φ by a large fraction. It’s important to use the anomalous value of Φ, so that the dilution rate is relative to an anomaly value of zero, which in this case is what we will consider the state of total dilution. Otherwise the dilution rate would describe the timescale to reach absolute zero, which for many quantities does not make physical sense, such as temperature.
An interesting aspect of the dilution rate is that it will go to zero if the air being entrained is the same as the air already in the cloud. This tends to make the dilution higher at upper levels in the atmosphere where the air tends to be drier, even though the entrainment tends to be weaker.
We can adapt the dilution rate to describe the dilution by any process. For example, we could estimate the dilution of cloud water by rainfall if we knew precisely how rain was falling out of a cloud. Unfortunately, these are all quantities that we can’t measure in the real world, so this technique is only really useful in high resolution cloud simulations. Luckily this tool can still help us learn more about entrainment and convection.
Dilution Results
I’ll just summarize a couple of the important findings from the paper, using total water (qt) for the dilution variable Φ. The results are from several simulations of individual clouds created by releasing a warm bubble in the SAM cloud model (Khairoutdinov and Randall, 2003).
The plot below shows the average profiles of dilution by different processes for the cloud core (i.e. strong updraft region), which I also refer to as “proportional tendencies“. The left panels shows the net dilution as the clouds are rising. The two middle panels show dilution by entrainment and detrainment, and the right panel shows the dilution by everything else, like precipitation fallout and turbulence.
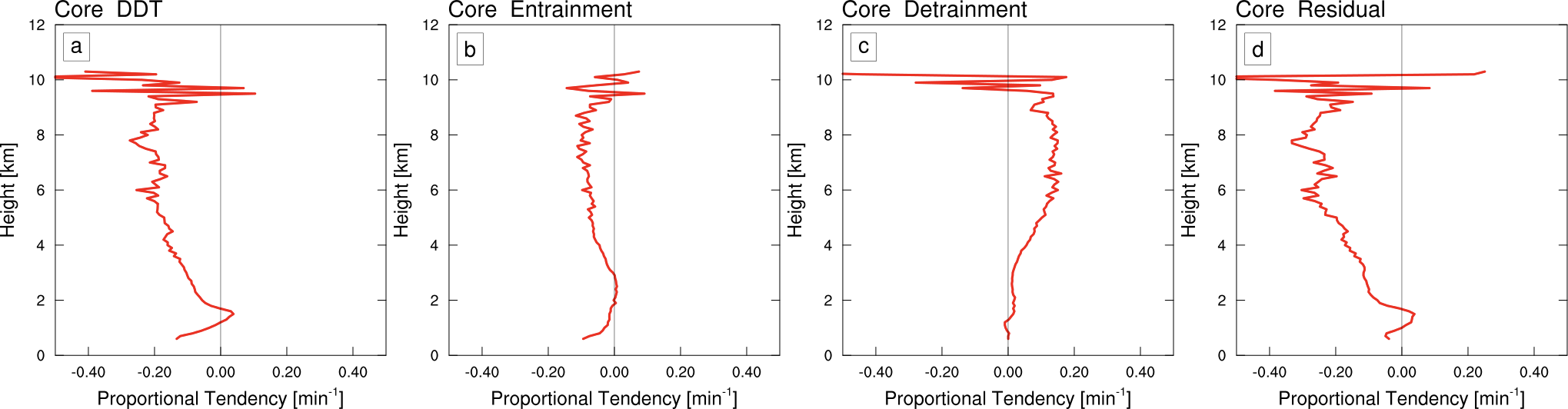
There are a few interesting things about this figure. First, entrainment has a negative tendency, which means it dilutes the cloud as we expect. However, I was surprised to find that detrainment actually plays a large role in concentrating the cloud water, which counteracts the dilution by entrainment. I think the explanation for this is that drier air is preferentially detrained, which leaves the cloud more humid on average. Since entrainment and detrainment tend to balance each other out, then the main factor diluting the cloud turns out to be all the other stuff like rain falling to the ground! I was pretty surprised to find this.
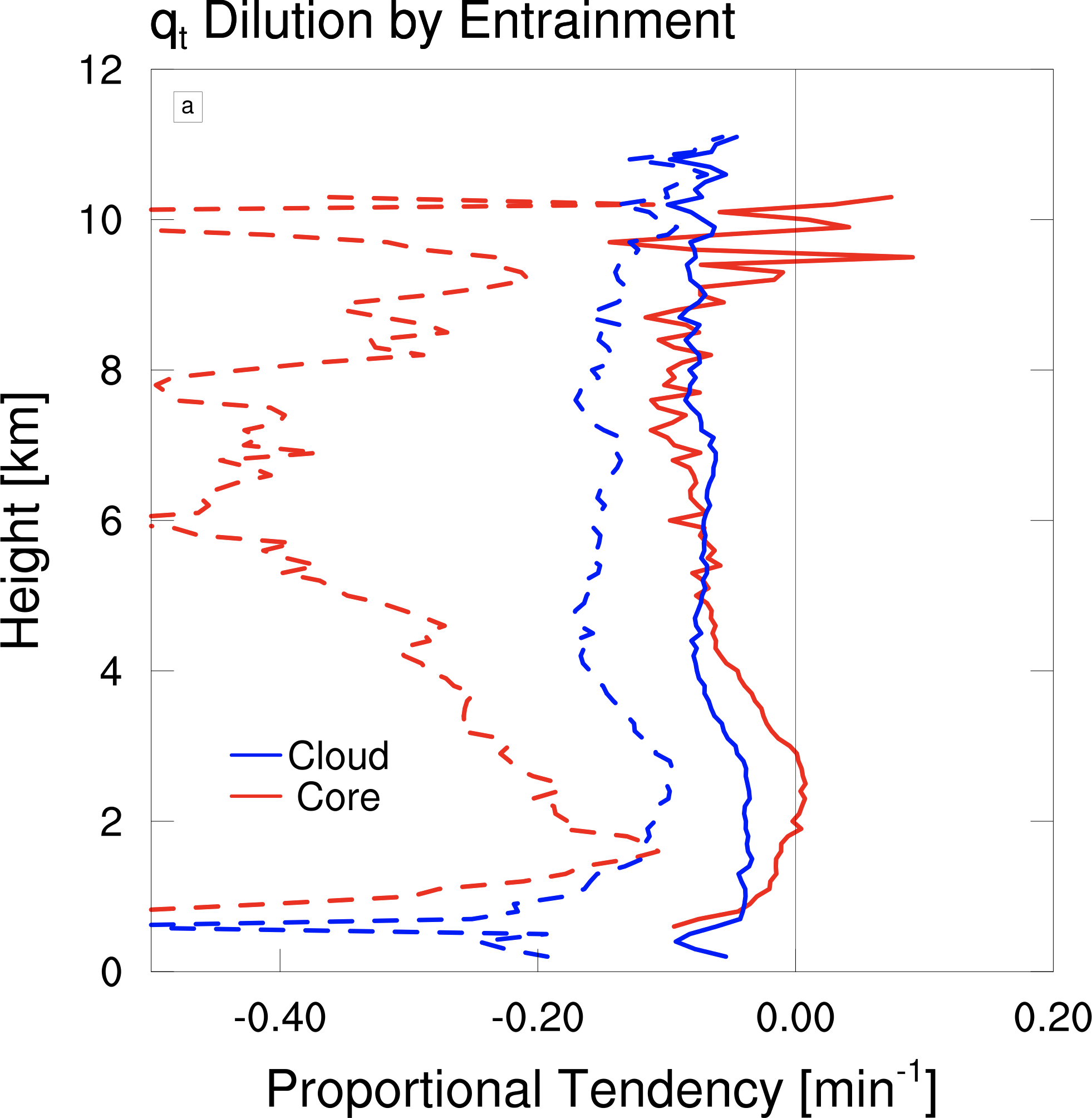
Another interesting thing we can do with the dilution rate is to calculate a hypothetical dilution rate that would occur if the air being entrained were different. Normally, air is entrained near the edge of a cloud, which may or may not have been influenced by the cloud already. The air may have also been influenced by a previous cloud. However, in a typical cumulus parameterization it is assumed that the air being entrained is from the average environment, which is dry because it is mostly devoid of clouds.
In the figure below I’ve plotted the dilution rate in solid lines for the cloud core (red) and the entire cloud volume (blue). The dashed lines show what those dilution rates would be if the entrained air came from the average air at each level. These hypothetical dilution rates are much higher than the actual dilution rates, and this difference is especially dramatic for the cloud core. This shows that the cloud is entraining air that tends to be much more humid than the environment. This also suggests that the moist air being entrained into the core has previously been in the cloud core, so there appears to be a large amount of recycling of cloud air, consistent with Yeo and Romps (2013).
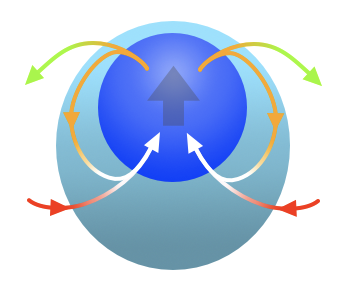
These results suggest that a moist convecting thermal might be more accurately described with a model that includes a shell of wet, non-buoyant air made up of air detrained by the thermal core, as shown in the cartoon below.

Congrats Walter….Interegsting
Can you pls send the pdf of the paper, no access to the pdf file from here…..
Thanks,
-Abhi
sent!